

A century after the invention of a technique to treat bacterial disease in humans with viruses that attack bacteria, a team at the Hebrew University has shown it to work. Groundbreaking research using a specific anti-bacterial virus against a specific bacterial pest on 16 “lost-cause” patients, treated on a compassionate basis produced a high 86.6 percent success rate: 13 of the 15 patients got better.
Indeed, a hundred years after the advent of “phage therapy,” it hadn’t been shown to work in an orderly manner in reproducible clinical trials, or been widely used to treat bacterial disease. It still hasn’t been, and isn’t. But now progress has been made and was reported in an international study led by two Israeli professors: Prof. Ran Nir-Paz at Hadassah Hebrew University Medical Center and Prof. Ronen Hazan of the Faculty of Dental Medicine at the Hebrew University.

Unleashing the nano-kraken
In 1915, as World War I raged, an English bacteriologist named William Twort discovered that bacteria in his lab had a problem. Two years later, the French-Canadian microbiologist Félix d’Hérelle independently realized what the problem was: bacteria could contract viruses, which he dubbed bacteriophages.
Thusly, Twort and d’Hérelle are credited with co-discovering the bacteriophage, the smallest of all viruses attacking the smallest of all hosts. Their discovery led to the emergence of a medical methodology – phage therapy – to treat bacterial infections of humans using bacteriophages.
In fact the French-Canadian microbiologist can also take the credit for the first attempts to harness these tiniest of parasites to vanquish pathogenic bacteria, in 1922, leading to efforts to develop phage therapy pretty much worldwide. Companies, especially in the United States, developed phages for therapy, but things did not go particularly well. Results were inconsistent and scientific validation was lacking.
And then came antibiotics, which were enormously easier to produce and administer. The new drugs went mainstream and almost put paid to phage therapy research – in the West.
In Eastern Europe, notably Georgia and Russia, efforts continued apace, explains Prof. Nir-Paz by phone. And now in 2023, the world is plagued by antibiotic-resistant bacteria and persistent, chronic infections that simply don’t respond to antibiotics (though the bacteria aren’t necessarily drug resistant – it’s a different thing), and phage therapy research is all the rage again, worldwide. Nir-Paz points out, though, that the research is partly motivated by romantic reasons, by which he means scientific curiosity.
In 2017, the sphere of phage therapy received a terrific boost from a paper originating in researchers from San Diego, reporting on the dramatic recovery of one Prof. Tom Patterson – a comatose patient suffering from multidrug-resistant Acinetobacter baumannii – after the application of experimental phage therapy (inspired by research in Tbilisi, Georgia).

Do note that phages are picky. A specific phage will parasitize only very specific bacteria. Luckily, Patterson’s doctors found three research teams with phages that target Acinetobacter.
Romantic or practical, in the paper published Tuesday in Med, the team reports “promising” success in applying a phage named PASA16 to treat persisting cases of a bacteria called Pseudomonas aeruginosa. This is a common germ in soil and water that can cause pneumonia, blood infection and more, mainly in the immunocompromised.
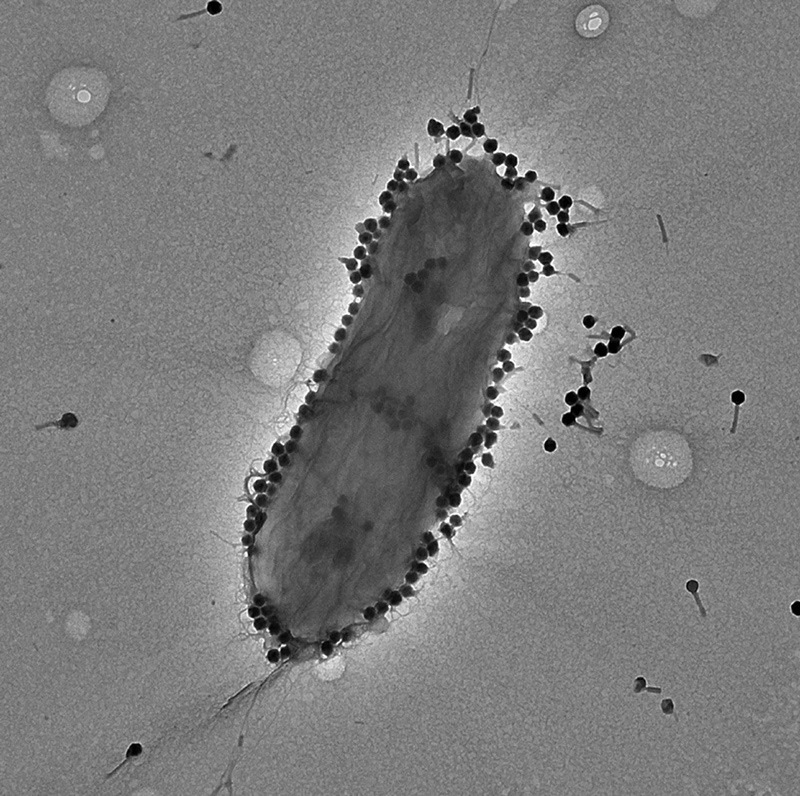
The treatment of the 16 untreatable patients suffering from persisting Pseudomonas infection was compassionate, in the sense that phage therapy has reached the stage of early phase-1 and phase-2 clinical trials, but no human testing yet. The 86.6 percent success rate supports the cause of further clinical trials in the case of Pseudomonas aeruginosa specifically, the team argues, which is where the new and groundbreaking aspect of the research lies, Nir-Paz explains.
To wit, so far phage therapy research has been vague in the sense of not focusing on one specific phage species attacking one specific bacteria species. This study does exactly that and is the biggest study of its kind so far. It concluded that PASA16 has potential to be the primary or auxiliary treatment of resistant or persistent Pseudomonas aeruginosa infection.
This is good news, if far short of a treatment coming to any hospital near you. At this point any phage therapy in Israel is compassionate, requiring specific permission per patient.
For each patient, treatment spanned from eight days to six weeks, with application of phage once or twice a day, intravenously or topically.
One reason that early-stage clinical trials have produced mixed results is because the researchers lacked the tools to figure out what to do in clinical trials in order to nail down the effect of treatment, Nir-Paz says. The new team achieved that and the 86.6 percent recovery rate.
One patient had a 20-year story of Pseudomonas leg infection; in despair, the thinking of his doctors was amputation. He is cured.
Another was en route to hospice care after a half-year of skin infection. She is healed. A girl with an infected leg also faced its loss, and today can run, skip and jump, Nir-Paz says.
There’s a long way to go, though. Prior to treatment, all Pseudomonas aeruginosa isolates from patients were tested and treatment was personalized. For instance, in one patient, the phage was shown to be effective in combination with antibiotic x. In another, it worked in combination with antibiotic y. In a third it didn’t work at all, so the third didn’t get the therapy. Not all who got the therapy were cured, he adds – two of their compassionate efforts did not work.
There could be many reasons why phage therapy might not work, he says. One possibility is that patients with persisting Pseudomonas already have the phage in their system and their bacteria are resistant to it too. So yes, phage-resistant bacteria is a thing as well.
Also, all this remains very expensive: for this study, the team was given free phages by the American phage production company Adaptive Phage Therapeutics.
“It’s still not clear in which direction this will go,” Nir-Paz sums up. There will be regulatory issues to tackle and, he points out, one cannot patent a phage. But he personally believes that based on their results – and others – compassionate treatment using phages will become more common, if one manages to overcome the financial hurdle. It’s not small.